New York City Zantac/Ranitidine Attorneys
Meirowitz & Wasserberg believe you deserve justice and compensation. Our experienced, defective pharmaceutical product attorneys offer free, private consultations to families who have been harmed by products like Zantac. Even if you aren’t sure your cancer was caused by Zantac, speaking with a lawyer and sharing the evidence of your case can help you better understand what happened as well as what to do next. To learn more, ask us a question, or schedule an appointment, please contact our offices by calling 212-897-1988 or filling out our short contact form.

Expertise
Mr. Wasserberg is proud to call himself a Trial Lawyer. He is often a featured speaker at industry summits and gatherings of the nation’s leading attorneys, from both sides of the bar. He is recognized by both his peers and his adversaries and is considered one of the nation’s premier mesothelioma and negligence attorneys
Content written by Dan Wasserberg

- Expertise
Mr. Wasserberg is proud to call himself a Trial Lawyer. He is often a featured speaker at industry summits and gatherings of the nation’s leading attorneys, from both sides of the bar. He is recognized by both his peers and his adversaries and is considered one of the nation’s premier mesothelioma and negligence attorneys
Experienced Mesothelioma Attorney
Quick Links
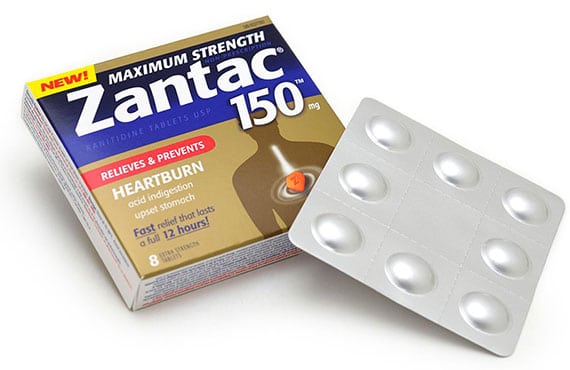
Every year, doctors write millions of prescriptions for ranitidine, better known as Zantac in its most popular version. The drug was at first available only by prescription, but is now available over the counter to treat heartburn, peptic ulcers, acid reflux, GERD and related conditions. Ever since its debut in 1981, it has been one of the best selling drugs in history – it has even been added to the World Health Organization’s Model List of Essential Medicines.
But in September of 2019, testing on Zantac showed the presence of a dangerous chemical called NDMA in several ranitidine products, at a level 3,000 to 26,000 times higher than the amount deemed by the FDA to be safe. NDMA is a known carcinogen (cancer causing agent), so potent that it has been used in medical studies to induce cancer in lab rats. Now pharmacies like CVS, Walgreens and Rite Aid are pulling Zantac and its generic versions from their shelves, and patients who have been diagnosed with cancer of the bladder, kidney, liver, uterus, stomach or esophagus after using Zantac are filing lawsuits against the manufacturers of the drug.
If you or a loved one has been diagnosed with cancer after using Zantac, the attorneys at Meirowitz & Wasserberg, LLP want to hear your story today.
What is NDMA, why is it in Zantac, and why is it is dangerous?
In September 2019 Valisure, an online pharmacy based in Connecticut, sounded the alarm to the FDA that their testing had discovered high levels of the dangerous chemical NDMA in several ranitidine products. NDMA (N-nitrosodimethylamine) has been medically classified as a probable carcinogen (cancer causing agent), which was at one point used in the production of rocket fuel. NDMA is the same carcinogen discovered in the blood-pressure medications Valsartan and Losartan, which led to their recalls in 2018. In that case, the drugs appeared to have been contaminated with NDMA during the manufacturing process by a drug ingredient factory in China. In Zantac’s case it’s theorized that, instead of Zantac becoming contaminated with NDMA during the manufacturing process, Zantac actually breaks down into NDMA during digestion due to its unstable nature. Further study is needed to confirm why NDMA is present in ranitidine (Zantac). According to the FDA, other anti-heartburn medications such as Pepcid (famotidine), Tagamet (cimetidine), Nexium (esomeprazole), Prevacid (lansoprazole), or Prilosec (omeprazole), have tested negative for NDMA.
Has the FDA recalled Ranitidine (Zantac)?
While the FDA has not issued a recall on Zantac and other ranitidine products, several manufacturers have issued voluntary recalls including Sanofi (manufacturer of brand name Zantac), and the FDA has been warning health care professionals and patients about these recalls. Walgreens, Walmart, Rite-Aid and CVS have begun taking Zantac and its generic versions off the shelves. On December 4, 2019, the FDA announced that it had manufacturers of Zantac and other ranitidine products to test ALL their products for NDMA before making them available to customers using testing methods developed specifically for this purpose by the FDA.
Effects of NDMA
Exposure to NDMA over time is potentially linked with the development of:
- Stomach or gastric Cancer
- Kidney Cancer
- Pancreatic Cancer
- Uterine Cancer
- Bladder Cancer
- Colon Cancer
- Intestinal Cancer
- Liver Cancer
- Esophageal Cancer
- Testicular Cancer
- Colorectal Cancer
If you or someone you know has used Zantac and been diagnosed with these cancers, you may have a case against the manufacturers of the drug. You may be entitled to compensation for:
- Your pain and suffering
- Your medical bills and related future medical bills
- Your lost wages and lost future earning capacity
- Your permanent disabilities resulting from the cancer and/or its treatment
- Your damaged quality of life
Free Confidential Consultations | Meirowitz & Wasserberg, LLP
Have you been diagnosed with any of the following cancers after using brand name prescription or over the counter Zantac for at least 60 days?
- Stomach or gastric Cancer
- Kidney Cancer
- Pancreatic Cancer
- Uterine Cancer
- Bladder Cancer
- Colon Cancer
- Intestinal Cancer
- Liver Cancer
- Esophageal Cancer
- Testicular Cancer
- Colorectal Cancer
If so, Meirowitz & Wasserberg believe you deserve justice and compensation. Our experienced, defective pharmaceutical product attorneys offer free, private consultations to families who have been harmed by products like Zantac. Even if you aren’t sure your cancer was caused by Zantac, speaking with a lawyer and sharing the evidence of your case can help you better understand what happened as well as what to do next. To learn more, ask us a question, or schedule an appointment, please contact our offices by calling 212-897-1988 or filling out our short contact form.
AVAILABLE 24/7
Get Your Free Case Review
Notable Results
$200,000
Personal injury settlement
$235,000
Slip and fall injury
$675,000
Product liability injury
$555,000
Premises liability injury
Nancy, New York (NY)






